This content was published in 2012. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Over 4% of 16- to 65-year-olds in England can be classed as alcohol-dependent and around a quarter of the population consumes alcohol in a way that is potentially harmful to health or well-being. It is a similar story for the rest of the UK: of the one million people in the UK addicted to alcohol only 6% per year receive treatment [1]. Possible contributing factors are under-identification of alcohol misuse by healthcare professionals, inability of people to identify they have a problem with alcohol and a lack of specialist treatment services.
Because of the high prevalence of alcohol misuse, most practising pharmacists will encounter the problem in some form. Accordingly, pharmacists need to have up-to-date knowledge of how to manage patients at risk of alcohol-related complications.
About alcohol withdrawal
Prolonged exposure to alcohol can cause adaptive changes in the brain’s receptors and neurotransmitters. These neurological effects manifest as tolerance, addiction and withdrawal. Alcohol is sedating, and so to maintain normal brain function down-regulation in inhibitory gamma-aminobutyric acid (GABA) receptors takes place, as does up-regulation of excitatory neurotransmitter receptors (including glutamine, serotonin and dopamine).
In short
The first step in managing alcohol withdrawal is to establish a patient’s drinking patterns. Pharmacological treatment can be used to prevent serious complications arising when the patient stops drinking.
Benzodiazepines are the treatment of choice and can be used in one of two regimens: a symptom-triggered approach or a fixed-dose regimen.
Abrupt cessation of alcohol removes the sedative effects of the alcohol but the adaptive changes in brain function persist. This leads to the characteristic alcohol withdrawal symptoms. The time it takes tore-establish normal brain neurotransmission determines the duration of the withdrawal.
Patients with chronic alcohol intake may stop drinking due to either an inability to obtain alcohol (because of lack of money, admission to hospital, illness, etc) or a desire to tackle their alcohol misuse. However, often patients who are withdrawing from alcohol do not wish to stop drinking. In these cases the aim of management is to prevent complications of withdrawal until they are able to obtain a drink.
Patients need to be motivated to stop drinking, as is the case for many drugs that have the potential for misuse. Pharmacological support for withdrawal is just one element of the overall treatment for alcohol dependence. There is some evidence that patients who have undergone multiple detoxification programmes are at an increased risk of withdrawal seizures and are less likely to maintain their abstinence.
Assessing the problem
Patients who have alcohol-related problems often find it difficult to accept they have a problem and have developed coping strategies to manage their drinking. It is essential for healthcare professionals to approach the treatment of this group in a non-judgemental and open way.
The first step in developing a dialogue is to establish patients’ drinking patterns, the impact on their lifestyle and their perception of the situation. There are many validated assessment tools that can help — from the complex “AUDIT” (alcohol use disorders identification test)through to the simple “CAGE” questions (see Box 1).
Box 1: Cage tool
The CAGE mnemonic is a simple tool for assessing people’s drinking patterns. Clinicians can ask patients the following four questions. Have you ever:
C…considered Cutting down your alcohol consumption
A…got Annoyed by people criticising your drinking
G…felt Guilty about your drinking
E…had a drink first thing in the morning (Eye opener)
Other things that need to be considered are concomitant health problems, other drug misuse, cognitive function and a person’s readiness for and belief in an ability to change. Patients who want to abstain from alcohol should be referred to specialist alcohol services to formulate and initiate a treatment plan.
They should be advised not to stop drinking alcohol abruptly, but can be given advice on gradual reduction to prevent the risk of withdrawal.
Pharmacological management
The pharmacological management of withdrawal is based on the same principles for patients wanting to stop drinking alcohol as for those who are unable to obtain alcohol. The aim of treatment is to prevent complications arising from the withdrawal. Withdrawal typically presents about six to 12 hours after ingestion of alcohol has stopped (correlating with reducing blood alcohol levels). Symptoms then tend to worsen, peaking at 48 to 72hours (see Box 2).
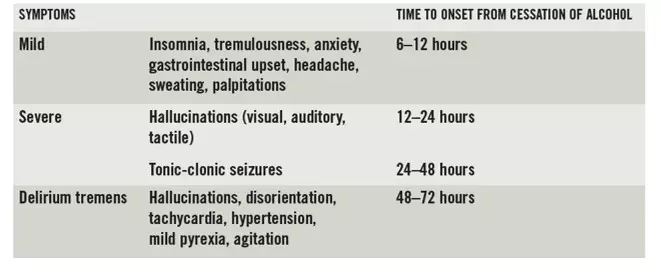
Delirium tremens is a serious withdrawal effect with a high mortality rate if not treated promptly; in the early 20th century mortality was as high as 37% but with modern medicine this has fallen to 5%. Patients need to have their alcohol withdrawal symptoms managed and be provided with adequate fluid and nutritional replacement. Controlling environmental stimuli is also beneficial in minimising the impact of hallucinations.
Chronic alcohol misuse can lead to B-group vitamin deficiency, which can cause adverse neurological effects; management of vitamin deficiency is discussed in Box 3.
Box 3: Vitamin B deficiency
Patients who have a history of chronic alcohol intake are also likely to be deficient in B-group vitamins. This is for a number of reasons, including:
- Inadequate diet
- Reduced absorption
- Increased metabolic demands
- Reduced hepatic storage
Thiamine (vitamin B1) deficiency can cause Wernicke’s encephalopathy (WE), which can ultimately progress to the irreversible Korsakoff’s psychosis.
Absorption of thiamine from the gastrointestinal tract is thiamine-dependent; therefore, patients deficient in thiamine are unlikely to absorb sufficient thiamine orally to prevent WE.
Patients therefore need to be given high-dose thiamine intravenously at the first possible opportunity in the form of Pabrinex (combination B vitamins). If there is no intravenous access then intramuscular Pabrinex is available.
Current best practice [4] is to provide doses of thiamine towards the upper end of the range set out in the British National Formulary. Once thiamine stores have been replenished, the patient can be switched to oral thiamine therapy (usually after five to seven days), which should be continued for as long as the patient is at risk of WE. A maximum of 5mg of thiamine can be absorbed from any one oral dose, so divided doses of thiamine are best; this needs to be balanced against the patient’s likelihood of adhering to therapy.
Benzodiazepines are the treatment of choice for alcohol withdrawal symptoms. The benzodiazepines bind to GABAreceptors (mimicking the sedative effects of alcohol) and control psychomotor agitation associated with withdrawal. Gradual reduction of the benzodiazepine allows normal homeostasis of the brain’s neurotransmitters to resume.
There is no strong evidence to support the choice of one benzodiazepine over any other [2]. Choice tends to be influenced by the different pharmacology of medicines and the perceived benefits for individual patients. Chlordiazepoxide, diazepam and lorazepam are the benzodiazepines that tend to be used most often (see below).
Carbamazepine and clomethiazole can also be used for alcohol withdrawal but should only be considered for use in specialist centres for patients who are difficult to manage with standard treatments. Clomethiazole in particular should rarely be used because of a high risk of respiratory depression when it is combined with alcohol.
Chlordiazepoxide
Chlordiazepoxide has a slow onset of action and therefore has lower abuse potential. In principle it should produce smoother withdrawal because the peaks and troughs associated with a shorter half-life drug would be avoided. There is an argument that due to its metabolism in the liver it is more likely to accumulate inpatients with hepatic disease, leading to over-sedation.
Diazepam
Diazepam has a faster onset of action, which could be beneficial in managing patients rapidly but may increase the potential for abuse. It has a long duration of action, so will provide a smoother withdrawal. The choice between diazepam and chlordiazepoxide will be dependent upon how much supervision can be provided (to prevent abuse) and the type of withdrawal regimen used (see below).
Lorazepam
Lorazepam (unlicensed) has a shorter duration of action, so tends to be reserved for patients who metabolise benzodiazepines less effectively, like the elderly or patients with liver failure. Lorazepam is the only benzodiazepine with consistent absorption when administered intramuscularly.
Detoxification approaches
Alcohol detoxification can take place in a variety of settings. On the whole it is more effective if planned and the necessary psychological and social support is in place. For stable patients who have mild or moderate symptoms, detoxification can be provided by specialist community alcohol services. Admission to hospital for closer observation of the initiation of withdrawal treatment is best practice for patients who are at risk of developing delirium tremens or whose withdrawal symptoms are severe [3].
For vulnerable patients, such as the elderly, people with cognitive impairment, those aged under 18 years and those with poor social support, community detoxification is less likely to be effective so admission to hospital should be considered. Care must be taken to ensure that patients are not provided with large quantities of benzodiazepines unsupervised; prescriptions should be kept to a maximum of two to three days’ treatment.
There are two main options for benzodiazepine regimens: symptom-triggered and fixed-dose. The choice of regimen will be dependent upon care setting, patient characteristics and availability of specialist supervision.
Symptom-triggered approach
Symptom-triggered withdrawal has been shown to provide effective treatment with lower benzodiazepine doses and shorter duration than fixed-dose regimens. It does, however, require patients to be closely supervised by skilled, trained staff. Patients are assessed regularly using a validated tool such as the “clinical institute withdrawal assessment of alcohol scale, revised” (CIWA-Ar). When the CIWA-Ar score is elevated the patient receives a dose of benzodiazepine. This usually ranges from 10mg to 100mg of chlordiazepoxide depending on the patient’s symptoms and continues until withdrawal symptoms are no longer present.
Fixed-dose regimen
The other option for withdrawal is a fixed-dose regimen. This involves an initial assessment of the patient’s withdrawal, preferably with a validated tool such as the “severity of alcohol dependence questionnaire” (SADQ) score. The patient is then placed on a fixed benzodiazepine regimen, which is reduced over seven to 10 days. The patient will receive a dose regardless of the presence of withdrawal symptoms. This can lead to over- or under-treatment of the patient. Because less specialist monitoring is required, fixed-dose regimens tend to be the treatment of choice for community and non-specialist hospital detoxification.
Maintaining abstinence
Medicines such as acamprosate, disulfiram and naltrexone can be used to help reduce cravings or support abstinence. Patients with chronic alcohol problems often have medical comorbidities (for example, liver disease and depression) and other substance misuse issues. It is essential that these are addressed to maximise the chance of effective abstinence.
Abstaining from alcohol can be a long journey for individuals. Withdrawal from alcohol is the first step, but to maintain abstinence psychological and social support are essential.
David Gibson is lead clinical pharmacist at Darlington Memorial Hospital, County Durham and Darlington NHS Foundation Trust. E: david.gibson@cddft.nhs.uk
NOTE
Clinical Pharmacist PRACTICE TOOLS do not constitute formal practice guidance. Articles in the series have been commissioned from independent authors who have summarised useful clinical skills.
References
- National Institute for Health and Clinical Excellence. Alcohol use disorders: diagnosis, assessment and management of harmful drinking and alcohol dependence. February 2011. www.nice.org.uk/cg115 (accessed 9 August 2012).
- Amato L, Minozzi S, Vecchi S, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database of Systematic Reviews 2010, issue 3.
- NHS Clinical Knowledge Summaries. Alcohol —problem drinking — management. www.cks.nhs.uk/alcohol_problem_drinking (accessed 9 August 2012).
- National Institute for Health and Clinical Excellence. Alcohol use disorders: diagnosis and clinical management of alcohol-related physical complications. June 2010. www.nice.org.uk/cg100 (accessed 9 August 2012).


