Superdrug
Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Source: Superdrug
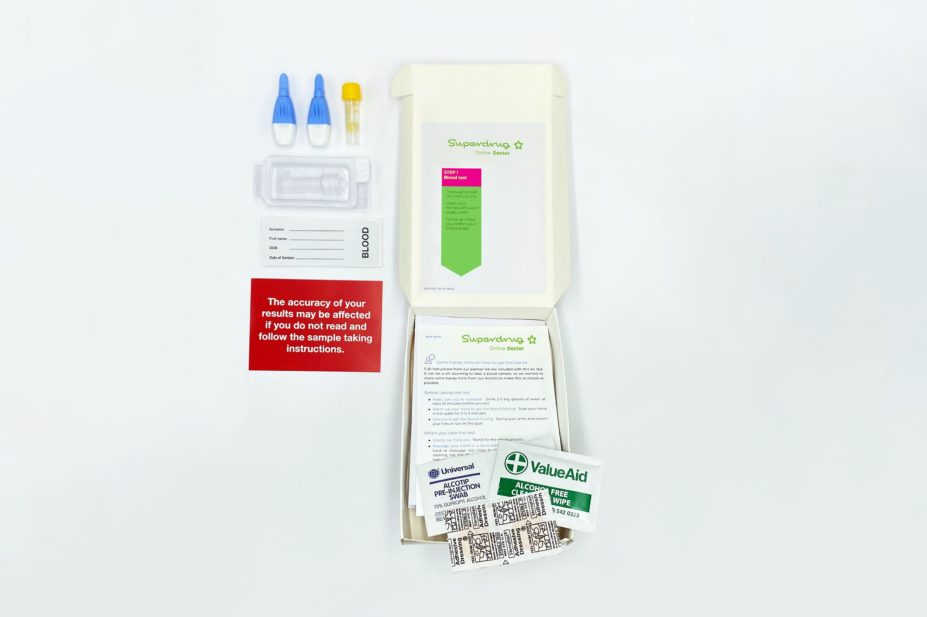
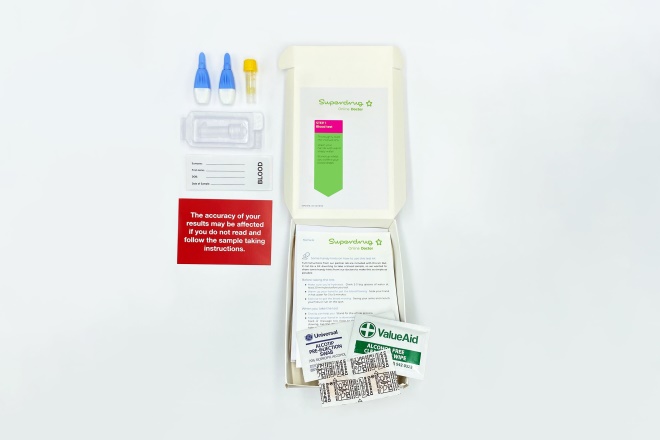
Superdrug’s COVID-19 antibody testing service involves a finger-prick blood sample, which is taken at home by the patient
High-street multiple Superdrug has relaunched its at-home COVID-19 antibody testing service, after it had been “voluntarily and temporarily” withdrawn in May 2020.
The antibody testing service, which is available at Superdrug Online Doctor for £69, involves a finger-prick blood sample which is taken at home by the patient and then sent to Superdrug’s partner laboratory for analysis.
According to Superdrug, the capillary blood sample is analysed using a Roche Elecsys Anti-Sars-CoV-2 test; the same assay being used for antibody analysis by the Department of Health and Social Care in adult social care. The test looks specifically for IgG antibodies.
Superdrug said the results are available “within 24 hours” of the sample reaching the lab.
The testing service was originally withdrawn after it was launched in May 2020 using a test manufactured by Abbott. The manufacturer had said that it had no evidence that the test could be carried out using a finger-prick blood sample.
In response to Superdrug’s latest announcement, Roche Diagnostics told The Pharmaceutical Journal that its Elecsys Anti-SARS-CoV-2 test complies with EU safety rules for serum and plasma taken by venipuncture or as capillary blood.
Commenting on the launch, Michael Henry, healthcare director at Superdrug, said: “Our team has worked hard over the last few months to offer a testing service that follows all of the regulatory guidelines.
“The decision to relaunch this service was driven by public demand because people want to know whether or not they have already been infected. In fact, our current data highlights that of those who have taken the test in the past 10 days, 13.5% have tested positive for COVID-19 antibodies.”
Guidance from the Medicines and Healthcare products Regulatory Agency notes that antibody tests can be purchased privately but adds that “it’s important to be aware that the General Pharmaceutical Council (GPhC) have advised pharmacies not to offer rapid antibody testing”, which provide results in about ten minutes.
In an email sent by the GPhC to stakeholders, and seen by The Pharmaceutical Journal, the regulator said it is taking action to “review our position in relation to rapid antibody tests, and other forms of COVID-19 testing, being provided in community pharmacies”.
“We are aware that this is a fast-changing landscape and there is significant interest across community pharmacy in being able to provide COVID-19 tests to the public,” the letter said.
“We have recently written again to the public health bodies, including Public Health England (PHE), to ask them to confirm their current position in relation to the provision of rapid antibody testing, rapid antigen testing and other forms of COVID-19 testing within settings such as community pharmacies.”
In May 2020, PHE published advice against the use of rapid point-of-care tests in community pharmacies.


