Ed / Wikipedia
Tardive dyskinesia is a term used to describe involuntary movements of the face or body and can be a side effect of chronic exposure to antipsychotics.
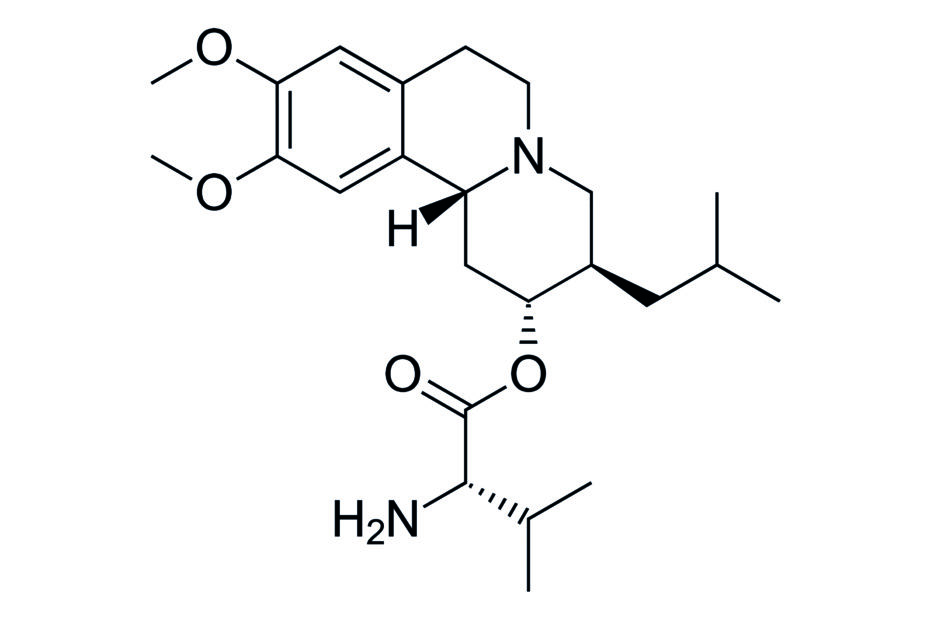
In a phase III study, researchers compared the efficacy of a novel treatment for the condition, valbenazine, at two different doses (40 and 80 mg/day), to placebo, in 225 people with moderate or severe tardive dyskinesia.
After six weeks, dyskinesia, as measured by the Abnormal Involuntary Movement Scale, was virtually unchanged in the placebo group at a mean of -0.1 units. A significantly greater decrease was observed in the active treatment groups at a mean of -1.9 units in the 40mg/day group and -3.2 units in the 80mg/day group.
Reporting in the American Journal of Psychiatry
[1]
(online, 21 March 2017), the team says the results indicate once-daily valbenazine could be an effective treatment for tardive dyskinesia. It was approved by the US Food and Drug Administration in April 2017.
References
[1] Hauser R, Factor S, Marder S et al. KINECT 3: A phase III randomized, double-blind, placebo-controlled trial of valbenazine for tardive dyskinesia. Am J Psychiatry 2017. doi: 10.1176/appi.ajp.2017.16091037.